NHS to Offer Weight-Loss Jab Wegovy for Heart Attack and Stroke Prevention
Wegovy to be Rolled Out on NHS
The National Health Service in England is set to introduce the weight-loss injection Wegovy, also known as semaglutide, for over a million people deemed at high risk of further heart attacks and strokes. The National Institute for Health and Care Excellence (NICE) has recommended the weekly jab as an “extra layer of protection” for individuals with established cardiovascular disease, including those who have previously suffered a heart attack or stroke, or have serious circulation problems in their legs, alongside being overweight or obese.
Clinical trials have demonstrated a compelling link between Wegovy and a 20% reduction in the risk of future heart attacks and strokes when administered in conjunction with existing cardiac medications. Notably, these benefits were observed early in the trials, suggesting the drug exerts direct positive effects on the heart and blood vessels, beyond just weight reduction.
Eligibility and Administration
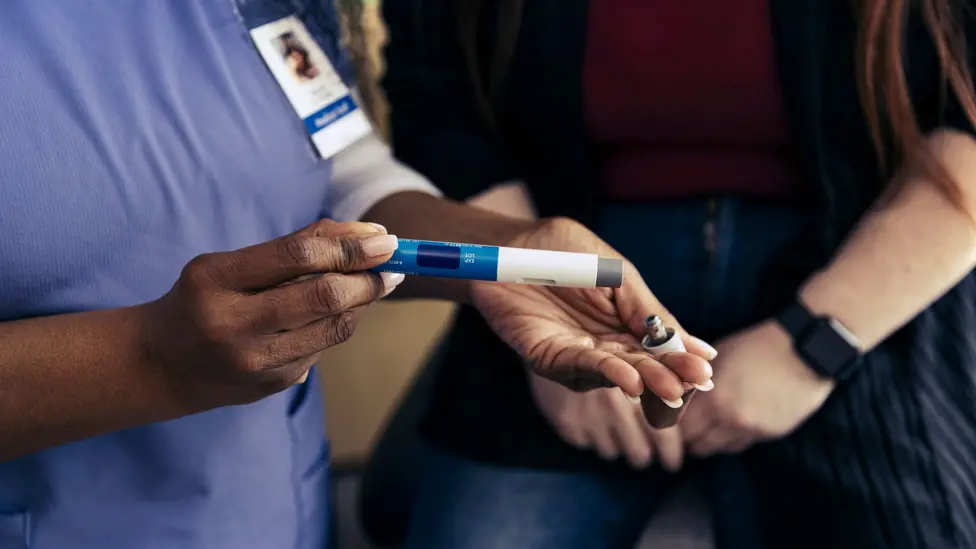
Patients with a Body Mass Index (BMI) of 27 or higher, classifying them as overweight or obese, will be considered for the treatment. A GP or specialist will determine suitability, especially for those already on other heart medications such as statins. The drug, which works as an appetite suppressant by mimicking the hormone GLP-1, will be self-administered at home via a pen injector device. Rollout is anticipated to commence this summer.
While Wegovy offers significant preventative potential, with experts estimating that around seven in ten cases of recurrent cardiovascular events could be averted, it is crucial to complement the treatment with healthy lifestyle changes, including diet and exercise. Concerns have been raised regarding potential side effects like bloating or nausea, and the importance of physical activity to counteract possible reductions in muscle mass.
Currently, NHS treatment with Wegovy is limited to two years via specialist services, with ongoing studies into its long-term effects. NICE has confirmed an agreement with pharmaceutical company Novo Nordisk to ensure the treatment is cost-effective for the NHS.

